Source #1 – Radon from Radium in Soils and Bedrock
Radium is a common element found in many types of soil and bedrock, and only a small amount is required for an indoor radon problem to develop. As a result, elevated radon levels have been found in every county of every state in the U.S. Before radon can enter the soil gas, the radium source must be near air or water in the soil pores. High soil gas concentrations are produced where a large portion of the radium is surrounded by air or water and there is little opportunity for dilution from atmospheric air.
Ranges of Soil Gas Radon Concentrations
Most soils are between 15 and 55 percent pore (void) space, which is sufficient for rapid transport. Weathered and fractured rocks such as exfoliated shale, glacier-ground granites, or water-eroded, mineralized limestone also have sufficient pore space for easy soil gas transport. Extreme variations have been observed in soil gas measurements, ranging from about 100 pCi/L to over 100,000 pCi/L. In one unusual example, soil measurements conducted for EPA radon study, taken within 30 feet of each other differed by a factor of 250%. The soil gas in one location had a radon concentration of 350 pCi/L, while the nearby location had 89,000 pCi/L (Ref. 6). This extreme variation makes it possible for similar buildings to be located right next to each other with very different indoor radon levels.
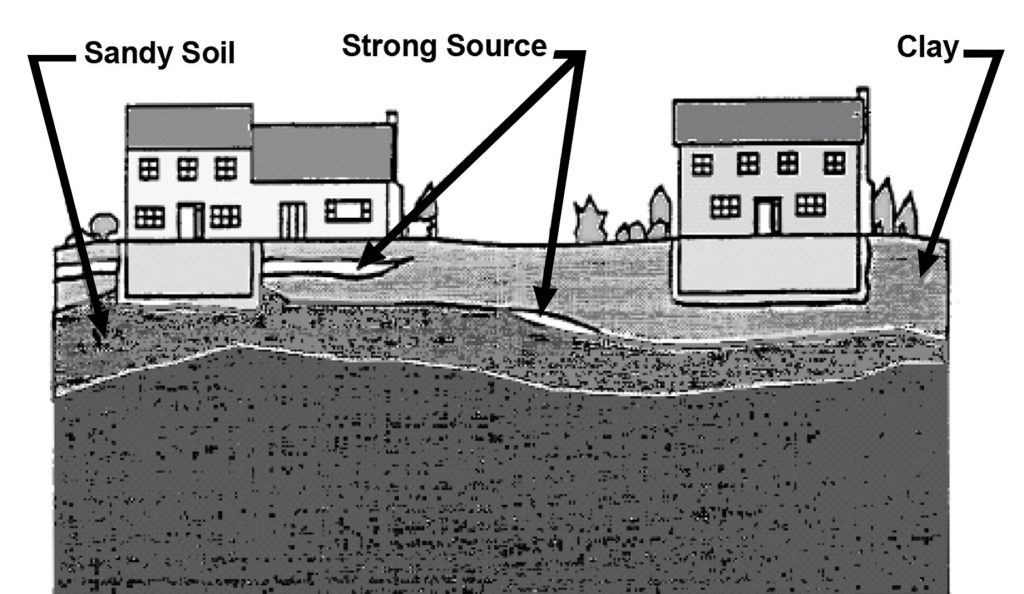
Radon Entry into Individualized Homes
Source: EPA
Figure 3-4 illustrates a hypothetical example of two adjacent buildings where radon concentrations would be expected to vary widely. One is protected from a strong source by a layer of clay, while the other has sandy soil at the base of its foundation in contact with the source. This type of situation illustrates why it is difficult to characterize the potential for radon problems prior to construction at a single building site.
